SCIENTIFIC REPORT
Project PN-III-P3-3.1-PM-RO-FR-2019-0165,
Contract no.: 18 BM/2019
“Efficient removal of
antibiotic pollutants in water by non-thermal plasma
coupled with other
advanced oxidation processes”
July - November 2019
1. Introduction
The project scope
is to develop an efficient method for the degradation of pharmaceutical compounds
in water, based on non-thermal plasma coupled with other advanced oxidation
processes (AOPs), such as ozonation and heterogeneous catalysis. The project
intends to combine experimental research and numerical simulations in order to achieve
deeper insight into the degradation of organic pollutants in water by
non-thermal plasma. The studies focus on antibiotics, since these compounds are
hardly biodegradable and difficult to destroy by conventional treatment
techniques and thus these highly persistent compounds are of greatest concern
regarding water contamination.
The
dramatic increase in the consumption of various medicines during the last years
is due to several reasons, such as the population growth, the inverting age
structure and the appearance of new target age groups. The discovery of new
drugs or of new uses for the existing ones also contribute to the increased
pharmaceuticals consumption. This leads to the high risk of water contamination
with these compounds. Harmful effects on aquatic and terrestrial organisms as
well as potential effects on human health made pharmaceutical pollutants to be
regarded as a major concern, especially keeping in mind the low
biodegradability in wastewater treatment plants observed for many of these
chemicals [1]. Antibiotics represent one of the most dangerous classes of
pharmaceuticals from the viewpoint of water contamination. The large-scale use
in human and veterinary medicine leads to the continuous introduction of these
compounds in the environment and explains the observed bioaccumulation and the
pseudo-persistent character [2]. Moreover, antibiotics can produce resistance
in bacterial populations [3], causing serious difficulties in the treatment of
various pathologies.
Conventional
water treatment techniques proved rather inefficient with respect to
antibiotics removal, especially in case of compounds presenting high toxicity
towards the microorganisms used in biological systems [2]. Therefore, research
focused more and more on alternative removal methods, generally known as
advanced oxidation processes (AOPs), such as ozonation, Fenton and photo-Fenton
processes, photocatalysis etc. Although ozonation proved effective for
antibiotics removal, it achieves very low mineralization and the resulting
degradation products are extremely toxic, as indicated by ecotoxicological
tests of the treated water [4]. Photo-Fenton oxidation appears as a more
promising method from the point of view of mineralization and biodegradability
of solutions containing antibiotics [3,5]. The governing principle of AOPs is
based on the formation of powerful oxidizing species, especially hydroxyl
radicals (OH), which react to the organic contaminants and degrade them.
Non-thermal
plasma also generates a large variety of oxidizers, including OH, and as an
advantage over the other AOPs, plasma produces the oxidizers in-situ, without
requiring the introduction of external additives [6]. Recently, the plasma
removal of several antibiotics has been reported [7-10], but the degradation
products are quite resistant and therefore the mineralization degree was
relatively low. The present work will be focused on enhancing mineralization
and increasing the degradation efficiency of antibiotics by combining
non-thermal plasma treatment with ozonation and heterogeneous catalysis.
In the first step
of the project a detailed characterization of the electrical discharge was
proposed, as well as the optimization of experimental conditions. A pulsed
corona discharge above water has been used, since this configuration already
proved efficient for the degradation of various organic contaminants in water
[11-14]. The effect of discharge characteristics, particularly the pulse
duration, on the removal of antibiotics from water has been investigated. The
influence of the gaseous atmosphere (oxygen, air) and of solution properties
(conductivity, pH, tap water vs. distilled water) has also been studied. The
evolution of the major degradation products has been followed. Preliminary
experiments comparing the results obtained by plasma alone and by
plasma-ozonation have been carried out.
2. Experimental
set-up
2.1.Plasma
reactor design
The experiments
were carried out using a pulsed corona discharge above water at atmospheric
pressure and room temperature. Oxygen or air were flown through the plasma
reactor with a flow rate of 300 mL/min. The plasma reactor (Fig. 1) is a
parallelepipedal vessel containing an aluminum ground electrode placed on the
bottom (Al tape) and the high voltage electrode consisting in an array of 20
copper wires (100 mm
diameter, 6 mm distance between adjacent wires) placed above the aqueous solution.
The distance between the wires and the ground electrode was approximately 10 mm
and the height of solution layer was 5 mm, so the discharge gap was around 5
mm. The discharge is filamentary, it starts at the wires and propagates towards
the surface of the liquid.
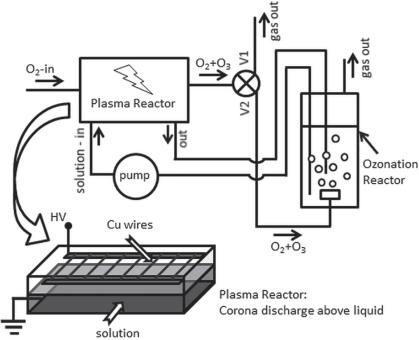
Fig. 1. Experimental set-up used for
plasma degradation of antibiotics [15]. The valve is on position V1 for plasma
alone and on position V2 for plasma-ozonation experiments
2.2.Electrical
circuit
The high voltage
pulses were generated using two different electrical circuits.
(1) by
using a capacitor of 6 nF charged at high voltage and
discharged by a solid-state thyristor switch (Behlke
HTS 240-800-SCR) triggered by a variable frequency pulse generator (ThurlbyThandar Instruments (TTI) TGP-110, 10 MHz) – Fig.
2(a).
(2) by
using a coaxial cable of 10 m length and a spark-gap switch operated in
self-breakdown mode to short-circuit the pulse forming line – Fig. 2(b).
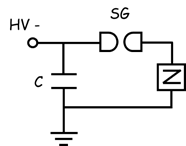 (a)
(a)  (b)
(b)
Fig. 2. Electrical circuits used for the
generation of high voltage pulses:
(a) capacitor-based circuit (C = 6 nF); (b) pulse-forming line circuit (C = 1.5 nF, R = … W).
HV – d.c. high
voltage power supply, SG – switch, Z – plasma reactor
Negative polarity
was used in all experiments. The voltage amplitude was 17-18 kV in both cases.
In most experiments the pulse repetition rate was maintained at 25 Hz, with one
exception where the pulse forming line circuit was used at 125 Hz, which will
be detailed in the results section.
The discharge
voltage and current were measured using probes (Tektronix P6015—voltage,
Tektronix TCP0150—current) and were monitored by a digital oscilloscope (Tektronix
DPO 2024). The energy per pulse was determined from the integration of the
current–voltage product over the pulse duration. The average power dissipated
in the discharge was calculated by multiplying the energy per pulse by the
frequency.
2.3.Solution
treatment configurations
The solution to be
treated (total volume 330 mL) is continuously circulated between the plasma
reactor and the solution reservoir (ozonation reactor) by a peristaltic pump,
with a flow rate of 160 mL/min. Most experiments were performed in plasma alone
configuration (valve on position V1), with the effluent gas from the discharge
vented immediately after the plasma reactor. For comparison, an experiment was
carried out in the plasma-ozonation configuration (valve on position V2), with
the effluent gas bubbled through the solution contained in the ozonation
reactor.
2.4.Solution
analysis
Several
antibiotics have been selected for the studies, according to various criteria,
such as the usage / consumption, the frequency of occurrence in the
environment, the biodegradability, the toxicity, the solubility in water etc.
The chosen antibiotics are listed in Table 1, together with their chemical
formula and chemical structure, as well as other relevant information. In the following
section the results obtained with amoxicillin will be presented in detail.
Solutions
containing the investigated antibiotics were prepared in distilled water in
most experiments. For comparison, some experiments were performed in tap water,
and this information will be mentioned where appropriate. The solution
conductivity was adjusted to the desired level with Na2SO4.
Two values of the conductivity were selected: 200 mS/cm and 500 mS/cm.
The initial pH of
solutions containing amoxicillin was 6.5. Experiments were also performed in
acidic pH, adjusted to 3.6 by H2SO4, and in basic pH,
adjusted to 10 by NaOH.
Table 1. Antibiotics selected for the
experimental investigations
|
Antibiotic |
Class |
Chemical structure,
formula, Molecular mass |
Solubility |
EU watchlist |
|
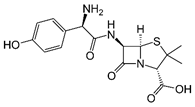
Amoxicillin |
penicillin |
C16H19N3O5S,
365.40 g/mol |
3.4 g/L @ 25 oC |
yes |
|
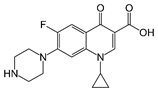
Ciprofloxacin |
quinolone |
C17H18FN3O3,
331.346 g/mol |
29.8 g/L |
yes |
|
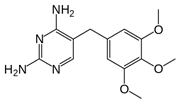
Trimethoprim |
|
C14H18N4O3,
290.32 g/mol |
610 mg/L @ 37 oC |
no |
|
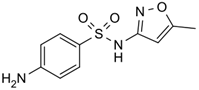
Sulfamethoxazole |
sulfonamide |
C10H11N3O3S,
253.279 g/mol |
400 mg/L @ 25 oC |
no |
|
Doxycycline |
tetracycline |
C22H24N2O8,
444.43 g/mol |
640 mg/L |
no |
The degradation of
amoxicillin was assessed using high performance liquid chromatography (HPLC),
employing a Rigol L-3000 system equipped with a
reverse phase C18 column (250 x 4,6 mm, particle size 5 mm).
The mobile phase consisted of acetonitrile (7%) and water with 0.1 % formic
acid (93%), at a flow rate of 2 mL/min. The oven temperature was set to 27 oC. Quantification of amoxicillin was done using
a diode array detector (DAD) set at a wavelength of 210 nm, while the 230 nm
was used for the specificity check. The peak UV-Vis spectrum was registered
between 210 and 410 nm.
3. Results
3.1.Electrical
characteristics of the discharge
Voltage and
current waveforms of the pulses used in the experiments are shown in Fig. 3 for
the solution containing amoxicillin (100 mg/L) of initial conductivity 500 mS/cm
and pH 6.5. As previously mentioned, the amplitude of the voltage was set to
17-18 kV.
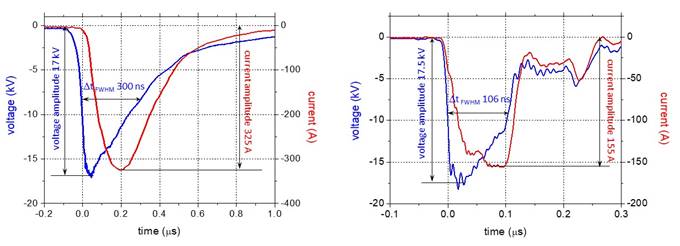
Fig. 3. Voltage and current waveforms of
the discharge pulses obtained with
the capacitor-based circuit (left) and
with the pulse forming line circuit (right)
(initial concentration of amoxicillin
solution 100 mg/L, initial conductivity 500 mS/cm, initial
pH 6.5)
The voltage pulses
obtained by discharging the capacitor have a typical shape (Fig. 3(a)), a rise
time of approximately 55 ns (determined by the switch characteristics) and a
duration (FWHM – full width at half maximum) of approximately 300 ns
(influenced by the solution conductivity). The amplitude of the current pulses was
325 A. The instantaneous power (I × V) during
the discharge pulse reached 4 MW. The energy per pulse (obtained by integrating
the instantaneous power over the entire pulse duration) was 1.1 J. The pulse
repetition rate for these experiments was 25 Hz, and therefore the average
power dissipated in the discharge was 27.5 W.
The waveforms
obtained using the pulse forming line based on the coaxial cable have shapes as
shown in Fig. 3(b). The rise time of the voltage pulses was 13.5 ns and the duration
(FWHM) was 106 ns. The pulse width depends on the length of the cable, being
twice the time needed for the wave to travel along the cable. Thus, for a 10 m
cable, the pulse width is around 100 ns, and is independent on solution
conductivity. The current amplitude was 155 A. The following values were
calculated from the current and voltage waveforms: instantaneous power 2.3 MW,
energy per pulse 207 mJ and average power 5.2 W.
3.2.Influence
of the pulse duration on the degradation of amoxicillin
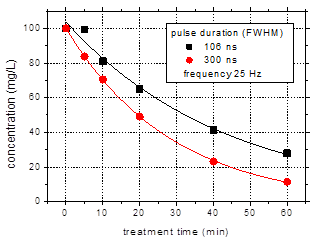
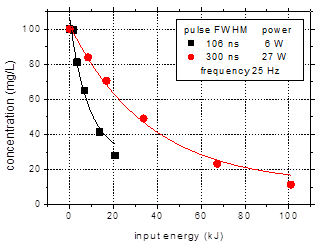
Fig. 4 shows the
results obtained with pulse durations of 106 and 300 ns with respect to the
degradation of amoxicillin (initial concentration 100 mg/L) in aqueous
solutions of 500 mS/cm
conductivity and pH 6.5. The experiments were performed for a pulse repetition
rate of 25 Hz.
 a
a
 b
b
Fig. 4. Degradation of amoxicillin
(initial concentration 100 mg/L) for pulse widths of 106 and 300 ns
as a function of: (a) – treatment time;
(b) – input energy.
(initial solution conductivity 500 mS/cm, initial pH 6.5)
The amoxicillin concentration
in the solutions exposed to plasma decreased exponentially with treatment time
(Fig. 4(a)). After 60 min treatment, 73% degradation of the antibiotic was
obtained using the shorter pulses, while in case of the 300 ns pulses the
removal was 89%. The faster degradation achieved with the longer discharge
pulses is due to the higher input power, i.e. 27 W, as compared to only 5-6 W
for the discharge operated with 106 ns pulses. For a more appropriate
comparison, the data were plotted as a function of input energy (Fig. 4(b)),
defined as the total amount of energy introduced in the plasma during water
treatment (i.e. the average power multiplied by the treatment time). Obviously,
the shorter discharge pulses are more efficient for the degradation of
contaminants, since the same values of removal are obtained for lower input
energy in case of the 106 ns pulses.
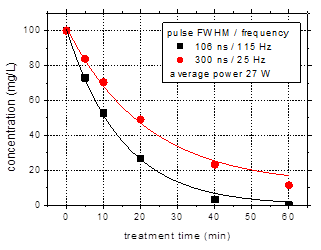
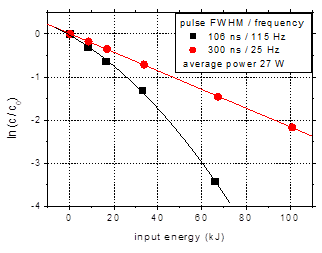
In order to
compare the results for the same average power, a series of experiments were
performed with the shorter pulses at higher frequency. Thus, the power
dissipated in the discharge is 27 W in both cases, i.e. 106 ns pulses at 115 Hz
and 300 ns pulses at 25 Hz. The results regarding amoxicillin degradation are
shown in Fig. 5.
 a
a
 b
b
Fig. 5. Degradation of amoxicillin
(initial concentration 100 mg/L) for pulse widths of 106 and 300 ns, at the
same average power (27 W) obtained by changing the pulse frequency (25 Hz / 115
Hz).
(a) concentration as a function of treatment
time; (b) logarithmic representation as a function of input energy. Initial
solution conductivity 500 mS/cm,
initial pH 6.5
Using shorter
pulses with higher repetition rate appears beneficial for the plasma removal of
antibiotic contaminants in water, leading to faster degradation of the
compounds (Fig. 5(a)). The entire amount of amoxicillin was eliminated during
60 min plasma treatment. The logarithmic representation of the data (Fig. 5(b))
shows a non-linear behavior in case of shorter discharge pulses, specifically
the acceleration of the degradation for long treatment time.
The efficiency of
the treatment method can be evaluated by calculating the energy yield, i.e. the
amount of pollutant removed per unit of energy introduced in the process. A
comparison from this point of view of the data presented so far is shown in
Fig. 6. The 300 ns pulses exhibit the lowest efficiency: at 50% conversion, the
energy yield was 1.9 g/kWh. Operating the discharge at the same power, but with
shorter pulses (106 ns) at higher frequency enhances the efficiency, in this
case the energy yield at 50% removal being 3.4 g/kWh. This effect is related to
the formation of reactive oxygen species (ROS) in the plasma and should be
correlated to the plasma characteristics and to the generation and consumption
mechanisms of these ROS [15].
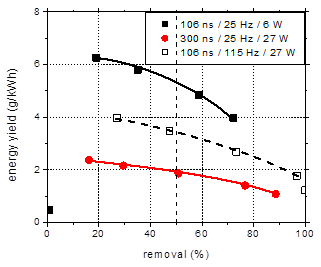
Fig. 6. Energy yield for the plasma
degradation of amoxicillin in water as a function of the removal rate.
Comparison between different discharge pulse widths.
(initial concentration 100 mg/L, initial
conductivity 500 mS/cm, initial pH 6.5)
Low power operation
with short discharge pulses at low frequency appears to be the most efficient
method for the degradation. At 50% amoxicillin removal the energy yield was 5.3
g/kWh, almost three times larger than the value obtained with the longer
pulses. However, the higher efficiency comes at the cost of slower degradation:
as previously mentioned, only 73% of the initial amoxicillin was removed after
60 min treatment at low power.
3.3.Influence
of the solution conductivity
The previous data
obtained for pulses of 106 ns at 25 Hz frequency and 500 mS/cm
initial solution conductivity are compared to results obtained for 200 mS/cm
in order to illustrate the effect of solution conductivity on the degradation (Fig.
7). The data are plotted as a function of input energy to account for the
slight difference in discharge power between the experiments (5.7–5.9 W). A
logarithmic representation is also shown (Fig. 7(b)) for a clearer picture of
the degradation behavior.
Faster removal of
the target compound was observed for lower solution conductivity. After 60 min
plasma treatment 88% of the initial amoxicillin from the 200 mS/cm
solution was degraded, as compared to 73% in case of the higher conductivity. Higher
solution conductivity (higher Na2SO4 concentration) could
affect the oxidative degradation of the target pollutant by: (i) diminishing the amoxicillin solubility in the aqueous
solution; (ii) more efficient interaction of sulfate ions with hydroxyl
radicals. Sulfate anions compete with the organic pollutants for the hydroxyl
radicals [16]:
HO‧
+ SO42- → SO4-‧ + OH-
However, the
resulted sulfate radical anions are strong oxidizer species. In the degradation
of amoxicillin, they have comparative reaction rate (2.9 ‧109 –
3.9 ‧109 M-1s-1) [17,18] with the
hydroxyl radicals (3.9 ‧ 109 – 6.9 ‧ 109 M-1s-1)
[18,19].
The solution
conductivity shows a slight increase during plasma treatment, reaching 521 mS/cm
and 245 mS/cm,
respectively, at the end of 60 min plasma exposure.
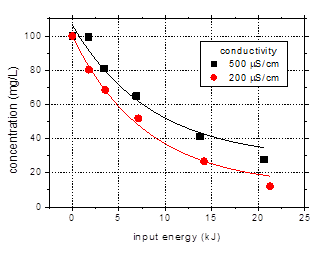 a
a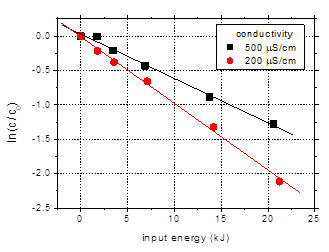 b
b
Fig. 7. Degradation of amoxicillin (initial
concentration 100 mg/L) as a function of input energy for solution
conductivities of 200 and 500 mS/cm
(initial pH 6.5, pulse width 106 ns, frequency 25 Hz)
3.4.
Influence of solution pH on the degradation of amoxicillin
The effect of
solution pH has been considered as well. Besides the previously mentioned results
obtained for pH 6.5, the degradation of amoxicillin in solutions of pH 3.6 and
10 have been investigated. The data plotted as a function of input energy as
well as the logarithmic representation are shown in Fig. 8.
The solution pH
decreases during plasma treatment due to the formation of acidic degradation
products. For the solution of initial pH 6.5, the final pH value, after 60 min
plasma exposure was 4.54. For the basic solution, the pH decreased to 6.73. On
the contrary, the acidic solution showed a slight pH increase as a result of
plasma exposure, the final value being 4.02.
Slightly faster
degradation of the target compound was observed in the acidic solution as
compared to the one of pH 6.5. In case of the basic solution, the removal is
considerably higher. After 60 min plasma treatment. 94.3% of the initial
amoxicillin was eliminated in the solution of initial pH 10, as compared to 73%
degradation in the solution of pH 6.5. The main reason of the higher removal of
the target compound at basic pH could be the increased rates of hydrogen
peroxide and ozone decomposition with formation of highly reactive hydroxyl
radicals [20,21]. Increased solubility of amoxicillin at basic pH (dissociation
constants: pK1 = 2.7 and pK2
= 7.2 – 7.4) [22] can also have a contribution to his faster removal
rate.
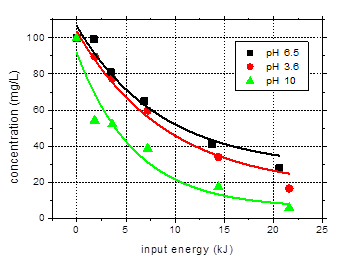
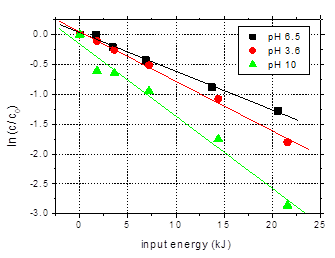
Fig. 8.
Degradation of amoxicillin (initial concentration 100 mg/L) as a
function of input energy for various solution pH: 3.6, 6.5, 10 (initial
conductivity 500 mS/cm, pulse width
106 ns, frequency 25 Hz)
3.5.
Comparison tap water vs. distilled water
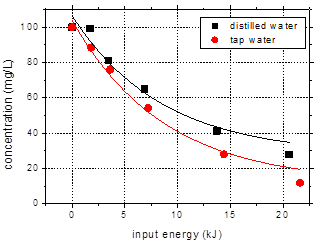
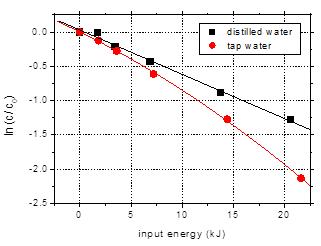
Tap water is a
useful model for the application of plasma treatment to wastewater and drinking
water purification, and therefore a comparison was made between the previously
mentioned data, obtained with distilled water, and results obtained for
solutions prepared in tap water (Fig. 9). The conductivity of tap water was 306
mS/cm
and the pH was 7.75. When adding amoxicillin into tap water the solution pH
became 6.84, while the conductivity was adjusted to 500 mS/cm
with Na2SO4. After 60 min plasma treatment the final pH
value was 7.35 and a slight increase in conductivity to 517 mS/cm
was observed. The carbonates present in tap water buffer the solution, so the
pH does not decrease during plasma treatment due to acids formation.
Although the
carbonate salts in tap water quench OH radicals [23] and thus should negatively
influence the degradation of organic compounds, the elimination of amoxicillin
was actually faster as compared to distilled water. After 60 min plasma
treatment the amoxicillin removal from tap water reached 88.2%, as compared to
only 73% in case of distilled water. This result can be explained by the
buffering effect of bicarbonate, which appears to dominate over adverse effects
due to the OH-scavenging character [24]. Obviously, this finding represents an
advantage in view of possible applications for water remediation.
 a
a
 b
b
Fig. 9. Degradation of amoxicillin (100
mg/L) in solutions prepared in distilled water and tap water as a function of
input energy (initial conductivity 500 mS/cm, pulse width 106 ns, frequency 25
Hz)
3.6.
Influence of the gaseous atmosphere
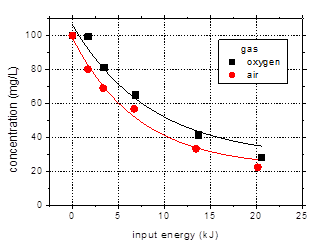
A comparison
between the experiments performed with plasma generated in oxygen and in air
has been made and the results regarding amoxicillin degradation are shown in
Fig. 10.
The pH drop in
case of the air plasma is more important than in solution treated in oxygen:
the final solution pH (after 60 min plasma treatment) was 3.71 for air, as
compared to 4.54 for oxygen. This is due
to the accumulation in the aqueous solution of nitrous and nitric acids, as a result
of reactive dissolution of nitrogen oxides generated in the air plasma system.
 a
a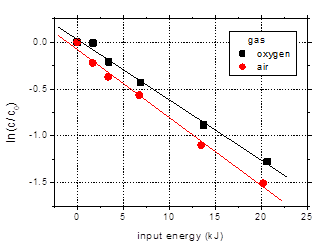 b
b
Fig. 10. Degradation of amoxicillin (initial
concentration 100 mg/L) by plasma generated in air and oxygen as a function of
input energy (distilled water, initial conductivity 500 mS/cm, initial pH 6.5, pulse
width 106 ns, frequency 25 Hz)
The influence of
the gaseous atmosphere on the removal of the target antibiotics appears to be
rather small under these conditions: 77.8% degradation in air as compared to
73% in oxygen after 60 min plasma treatment. This is not a significant
difference, so it can be said that both gases performed equally well. This is
another important result from the viewpoint of practical application of plasma to
the water remediation process.
3.7.
Comparison between plasma alone and plasma-ozonation
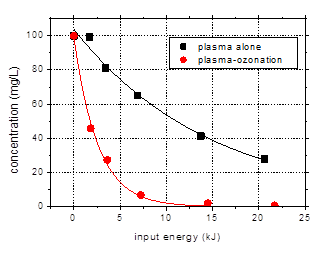
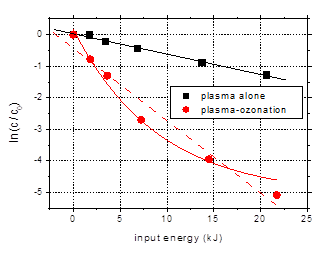
Preliminary
results achieved in the plasma-ozonation configuration are presented in Fig. 11
and compared with the data obtained by plasma alone. Here only a description of
the results is presented, while a through analysis makes the object of further
research that will be conducted in the next step of the project.
 a
a
 b
b
Fig. 11. Degradation of amoxicillin (100
mg/L) by plasma alone and plasma-ozonation as a function of input energy
(distilled water, oxygen plasma, initial conductivity 500 mS/cm, initial pH
6.5, pulse width 106 ns, frequency 25 Hz)
Plasma-ozonation
greatly enhances degradation, as clearly shown in the above figure. Amoxicillin
is almost completely degraded after 40 min treatment. After only 5 min
plasma-ozonation, more than 50% of the initial amoxicillin was removed. The
reaction rate constant was almost four times larger for plasma-ozonation than
for plasma alone. The deviation from linearity observed in the logarithmic
representation of the data (Fig. 11(b)) will need further study.
With respect to
the efficiency of the degradation process, the energy yield was 35.6 g/kWh for
plasma-ozonation, as compared to 5.3 g/kWh for plasma alone. The observed
degradation products are also eliminated in a large extent, indicating a high
degree of mineralization.
3.8.
Participation and tasks of the Romanian researchers
Dr. M. Nistor and Dr. D. Dobrin have scientific tasks mainly
related to the discharge electrical characterization: measurements of discharge
current and voltage under various experimental conditions and with various
pulse generation systems, calculation of energy and power etc.
Dr. C. Bradu and F. Bilea are mainly in
charge with the chemical characterization of plasma-treated solutions:
measurements of pH and conductivity, chromatographic analysis, identification
of degradation products, total organic carbon analysis etc. It is worth
mentioning that F. Bilea recently started his PhD
studies in the area of Analytical Chemistry, on a topic concerning the plasma
degradation of pharmaceutical compounds, therefore very close to the project
objective.
Dr. M. Magureanu
supervises the team activity and participates in all the experimental
activities.
3.9.
Common activities of the partners
Dr. Monica
Magureanu and Dr. Corina Bradu have visited GREMI
during the period 20-26 October 2019. They visited the various research groups
of the GREMI laboratory and discussed on the main topics of interest. During
this visit each of them presented a seminar:
Dr. C. Bradu: “Chimie de l’eau a Bucarest”
Dr. M. Magureanu:
“Degradation of organic pollutants in water by non-thermal
plasma” – web‑seminar broadcasted by Réseaux des Plasmas Froids
(http://plasmasfroids.cnrs.fr/spip.php?article365)
The kick-off
meeting of the project was also organized during this visit. The participants
shared ideas regarding the experimental research activities and their
correlation with the theoretical simulations, discussed the selection criteria of
the target compounds and decided on the antibiotics to be treated by plasma, and
finally discussed about the long-term visits of the two PhD students (Noussaiba Korichi and Florin Bilea) that will take place in 2020. Discussions about
common publication(s) that will include the results of the Romanian team
(presented above) and the results of the French group have also taken place.
Dr. Hervé Rabat and Dr. Olivier Aubry
have visited NILPRP during the period 10-15 November. During this visit, each of
them presented a seminar:
Dr. H. Rabat “From
an overview of the rreaserch at the GREMI laboratory
to a focus on the different researches in the Arc Team”
Dr. O. Aubry “Pharmaceutical molecules degradation in water by
non-thermal plasma and heterogeneous catalysis coupling”
The French
researchers visited several research groups from the Plasma Physics and Nuclear
Fusion Laboratory: the Plasma Chemistry and Advanced Functional Materials
Group, the Plasma Surface Engineering Group and the X-ray Imaging Laboratory:
Microtomography and Microfluorescence. They have also
visited the PROTMED research center of the University of Bucharest (Research
Center for Environmental Protection and Waste Management).
References:
[1] S.K. Khetan and T.J.
Collins, Chem. Rev. 107, 2319-2364, 2007.
[2] V. Homem and L. Santos,
J. Environ. Manag. 92, 2304-2347, 2011.
[3] A.G. Trovó et al., J. Photochem. Photobiol. A 198,
215-220, 2008.
[4] R. Andreozzi et al., J.
Hazard. Mater. 122, 243-250, 2005.
[5] E. Elmolla and M.
Chaudhuri, J. Hazard. Mater. 170, 666-672, 2009.
[6] P. Lukes et al., In: Parvulescu, V.I., Magureanu, M., Lukes, P. (Eds.), Plasma Chemistry and Catalysis in Gases
and Liquids. Wiley-VCH, pp. 243-308, 2012.
[7] M. Magureanu et al., Water Res. 45, 3407-3416,
2011.
[8] S.-P. Rong and Y.-B. Sun, J. Chem. Technol. Biotechnol. 89, 1351-1359,
2013.
[9] S.-P. Rong et al., Chin. Chem.
Lett. 25, 187-192, 2014.
[10] D. He, et al., Chem. Eng.
J. 258, 18-25, 2014.
[11] D. Dobrin, C. Bradu, M. Magureanu, N.B. Mandache, V.I. Parvulescu, Chem. Eng. J. 234, 389, 2013
[12] D. Dobrin, M. Magureanu, C. Bradu, N.B. Mandache, P. Ionita, V.I. Parvulescu, Environ. Sci. Pollut. Res. 21, 12190–12197,
2014
[13] C. Bradu, M. Magureanu, V.I. Parvulescu, J.
Hazard. Mater. 336, 52-56, 2017
[14] M. Magureanu, N.B. Mandache, C. Bradu, V.I. Parvulescu, Plasma Process. Polym.
15, e1700201, 2018
[15] F. Bilea, C. Bradu, N.B. Mandache, M. Magureanu, Chemosphere 236,
124302, 2019
[16] F. Ghanbaria, M. Moradib, F. Gohari, J. Wat. Proc.
Eng. 9, 22–28, 2016
[17] L. Wojnárovits, E. Takács, Chemosphere 220, 1014-1032, 2019
[18] S. P. Mezyk, K. A.
Rickman, G. McKay, C. M. Hirsch, X. He, D. D. Dionysiou, Aquatic Redox
Chemistry: Chapter 12, 247-263, 2011
[19] R. Andreozzi, M. Canterino, R. Marotta, N. Paxeus,
J. Haz. Mat. 122, 243-50, 2005
[20] Y. S. Junga, W. T.
Lima, J-Y. Parkb, Y-H. Kim, Environ. Technol. 30,
183–190, 2009
[21] B. G. Ershov, P. A.
Morozov, Russian J. of Appl. Chem., 81, 1895–1898, 2008
[22] I. M. Bezerra1, O. Chiavone-Filho,
S. Mattedi, Brazilian J.
Chem. Eng. 30, 45 – 54, 2013
[23] J. Hoigne, H. Bader,
Water Research 10 (5), 377-386, 1976
[24] E. Marotta, E. Ceriani,
M. Schiorlin, C. Ceretta,
C. Paradisi, Water Res. 46, 6239-6246, 2012